Biologists use microfluidics chips to watch worm behavior
Tiny roundworms called Caenorhabditis elegans have a rather uncomplicated method for finding food: They wriggle and turn and explore new territory until they find something edible, and then they stay the course until the food disappears. But despite the worm’s simple nervous system, researchers have been unable to figure out exactly how a stimulus such as food odor gets converted into a more complex, long-lasting behavior like foraging.
Now, using tools borrowed from the computer industry, Rockefeller University researchers have created tiny devices that allow them to watch in real time as information travels from one worm neuron to another. Their findings — that one of the long-studied olfactory neurons has a totally different mode of operation than expected — uncover a surprising similarity between the worm’s neural network and our own.
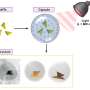
C. elegans has just 302 nerves in all, 32 of which are dedicated to smell. Such a basic system makes the worms ideal for investigating relationships among neural connections, genes and behavior. But because the microscopic creatures squirm in all three dimensions — up, down and side to side — scientists have had a hard time keeping them from wriggling right out of focus. So the scientists put the worms on chips. Cori Bargmann, Torsten N. Wiesel Professor and head of the Laboratory of Neural Circuits and Behavior, and postdoc Nikos Chronis built tiny microfluidics chips that let them introduce a single worm to minute, controlled amounts of fluid and provided a way to watch its behavior and neural reactions.
In two papers published this fall, the researchers and their colleagues describe how they created and used two different chips: one to measure olfactory neuron activity, and one to measure neural activity in relation to behavior. Both chips keep the worm restrained in order to keep it in focus: The behavior chip is just large enough for a single worm to move forward and backward, while the olfactory chip keeps a worm contained in a narrow space with just the tip of its nose sticking out into a controlled odor stream. “We’ve been having a lot of fun just figuring out what you can and can’t do,” Bargmann says.
One of the things they could do was monitor C. elegans’ neural activity in real time — the first time that had been achieved in the creature’s olfactory system. Then, once they knew that the two chip techniques worked, the researchers applied them to following the neural path from a stimulus to behavior. With postdoc and coauthor Sreekanth Chalasani, Bargmann and Chronis focused on one of the worm’s dominant olfactory neurons, called the AWC, and their results show that the neurons work nothing like they expected.
“We think of these neurons as olfactory neurons, but when we gave them odors, they did nothing,” Bargmann says. “And we gave them more and more odors and they did a whole lot of nothing. And after what seemed like years of frustration, we realized that when you took odors away, they responded.” The neurons, it turns out, are instead dedicated to food-seeking: They become activated when food odor disappears, and remain active until the odor returns.
The amount of activity of the neuron depends on how strong the odor is, and how long it lasts. “We can see the transformation of previous history into a single cell’s response,” Bargmann says. “So somewhere in the background, while it’s apparently silent, it’s actually noticing how long it’s been silent and using that to calibrate how big a response it will generate when the odor gets taken away.”
The neuron itself is only active for about 30 seconds. But the worm continues its search behavior for long after the activity has disappeared, which prompted the scientists to look at the neuron’s downstream targets. There, they could see that the AWC’s activity was essentially flipping a switch, turning the target neuron on and keeping it on for as long as they watched. How that causes the worms to double-back and turn in circles to seek food, “we can’t say yet,” Bargmann says. “But we’re really beginning to understand the relationship between neurons and behavior in a way that reflects their dynamics.”
Even more significantly, the pattern found by Bargmann, Chalasani and Chronis — who’s now an assistant professor of mechanical engineering at the University of Michigan — has an equivalent in the rod- and cone-shaped cells of the vertebrate eye. These cells are inactive in the light, and strongly activated when the lights go off. Even the molecules that regulate these actions are the same, or similar, to the ones that regulate them in C. elegans. “It’s an analogy,” Bargmann says. “But there may actually be some basic conserved components.” And these components might be the building blocks for particular kinds of neural connections, no matter what species they’re found in.
References:
Nature 450(7166): 63–70 (November 1, 2007)
Nature Methods 4(9): 727–731 (September, 2007)
Source: Rockefeller University