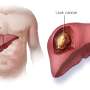
Tiny radioactive spheres effectively treat cancer that has spread to the liver
Placing tiny radioactive spheres directly into the liver through its blood supply halted growth of tumors that had spread to the organ in 71 percent of patients tested in a small clinical trial, researchers from Mayo Clinic Jacksonville report.
They say that the technique appears to offer a treatment option for patients who develop multiple tumors in their liver from cancer metastasis.
“Most of these patients don’t have other effective treatment options, because surgery is not possible if there are multiple tumors in their liver,” says the study’s lead investigator, Laura Vallow, M.D. “But with this radiotherapy, no new tumors developed in patients who responded and we find this to be very encouraging.”
Vallow presented results of the study at the annual meeting of the American Society for Therapeutic Radiation and Oncology (ASTRO).
She says her report is one of the first to sum up an initial clinical experience with the therapy, known as SIR-Spheres microspheres, which was approved for use by the federal Food and Drug Administration in 2002. “We are trying to determine the clinical situations in which it is best to use these microspheres,” Vallow says.
The therapy uses millions of tiny polymer beads − each one of which is about one-third the diameter of a human hair − which contain the radioactive element yttrium-90. An interventional radiologist uses a catheter to infuse the microspheres directly into the hepatic artery, which supplies all the blood to the liver. There the beads deliver radiation directly to tumors for about 11 days.
In this study, data from 20 patient procedures using microspheres at Mayo Clinic Jacksonville were analyzed. The entire liver was treated in 75 percent of the cases, and in the remaining 25 percent, either the left or right lobe of the liver was selectively targeted. Four weeks after treatment, researchers used CT scans to assess response of tumors, and found that in 71 percent, tumors decreased in size. “The tumors were smaller or less active with less contrast uptake on follow-up CT scans,” Vallow says.
The majority of patients had minimal side effects from the treatment, she says. Cancer progressed in two patients and they died within three months. But for the patients who responded, by the end of a 10-month follow-up period, no new tumors were detected, Vallow says. “Liver function tests in the responding patients have become normal or have stabilized.”
The researchers say the treatment works best in patients that have good blood flow to their tumors. “Further evaluation with longer follow-up will help clarify the benefit of this novel treatment,” Vallow says.
Source: Mayo Clinic