Power-boosting signal in muscle declines with age
As people age, they may have to exercise even harder to get the benefits afforded to younger folk. That's the suggestion of a report in the February issue of the journal Cell Metabolism, published by Cell Press, showing that a signal that gives muscles a kind of metabolic boost in response to exercise is blunted in older animals.
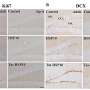
The researchers found that so-called AMP-activated protein kinase (AMPK) slows down in the skeletal muscle of 2-year-old rats relative to 3-month-old rats. A chief regulator of whole-body energy balance, AMPK in skeletal muscle stimulates the oxidation of fatty acids and the production, or biogenesis, of power-producing mitochondria that burn fat and fuel cells, according to the researchers.
The new findings might help to explain "what happens as we age," said Gerald I. Shulman, a Howard Hughes Medical Institute investigator at Yale University School of Medicine.
Earlier studies have shown that, in comparison to 20-year-old adults, even lean and healthy people in their seventies show a higher incidence of fat buildup in their muscle and livers and a deficiency in mitochondrial function, Shulman explained. These metabolic shifts have been implicated in the increased prevalence of insulin resistance and type 2 diabetes that occurs with aging.
"The message of this paper is that, with aging, the AMPK pathway has reduced activity," Shulman said. "So, one probably has to work harder to maintain the same level of fat oxidation and mitochondrial biogenesis in muscle."
The number of mitochondria within muscle largely determines its metabolic capacity. The mitochondrial composition of fibers in different muscles corresponds to the jobs that they do, and muscle can be "reprogrammed" through exercise.
For example, Shulman said, the skeletal muscles of marathon runners typically have much greater mitochondrial content and a greater capacity to burn fat. Those properties can most likely be attributed to chronic activation of the AMPK pathway.
"In regards to insulin resistance and type 2 diabetes, having more AMPK activity in our skeletal muscle is probably a good thing because AMPK activation stimulates glucose uptake, increases fat oxidation, and promotes mitochondrial biogenesis," he said.
In the current study, the researchers set out to determine whether the declining mitochondrial function and increased intracellular fat content seen with aging could be traced back to deficiencies of AMPK. They compared AMPK activity in young and old rats following three "perturbations" that normally stimulate the enzyme and, in turn, mitochondria production. The treatments included acute exposure to an AMPK-stimulating chemical, chronic exposure through feeding of another chemical that induces AMPK by mimicking an energy shortage, and exercise.
In every case, older rats showed a decline in AMPK activity compared to younger animals. Young rats infused with a stimulatory chemical showed an increase in muscular AMPK activity not seen in old rats, they found. Similarly, the muscle of exercise-trained young rats showed more than a doubling in AMPK activity. In older rats, that AMPK hike with exercise was "severely blunted." The muscles of young rats fed the AMPK-stimulating chemical also showed an increase in AMPK and a 38% increase in mitochondrial density, they reported. In contrast, older animals' AMPK activity and mitochondrial numbers held steady.
"These results suggest that aging-associated reductions in AMPK-stimulated activity may be an important contributing factor in the reduced mitochondrial function and dysregulated intracellular fat metabolism associated with aging-induced insulin resistance and type 2 diabetes," the researchers concluded.
Source: Cell Press