June 9, 2006 feature
Nano-tip could play integral part in heat-assisted data storage devices
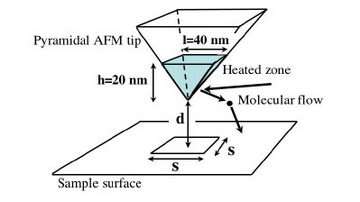
Using a tip with a nano heat source that never touches the surface, scientists have shown how to heat a localized surface with no contact. The discovery could open the doors to heat-assisted data storage devices and nano thermometers.
Every year, the world’s data storage needs more than double. Understanding heat transfer on the nanoscale is imperative for fabricating technology that will affect nearly everyone living in first-world countries. All over the world, scientists are rushing to develop an alternative data storage system in an effort to increase the space that our information-overloaded society is running out of.
One of the options, called thermally-assisted or heat-assisted data storage, works by using a laser to heat a data disc, which stabilizes the magnetic recording process by making it easier to write data when hot and retain data when subsequently cooled. The process overcomes a critical point that conventional magnetic recording devices will soon encounter, known as the superparamagnetic limit. As scientists increase data storage on conventional systems by making the room-temperature bits smaller, at a certain size the bits will become magnetically unstable, will fall out of place, and their information will vanish.
In a recent study on heat transfer between a tip and a surface, scientists from France have made a significant step toward the development of heat-assisted data storage, as well as other applications. The team calculated the heat transfer between a silicon tip and a surface, which is dominated mostly by air conduction.
“Heat transfer is well known at the macroscopic level (described by Fourier diffusion when collisions between molecules induce thermodynamic local equilibrium),” Pierre-Olivier Chapuis, coauthor of the recent paper in Nanotechnology, told PhysOrg.com. “Heat transfer can also be calculated in the pure ballistic regime (when there is no collision between molecules). But calculating heat transfer in the intermediate regime, when there are a few collisions, still remains a challenge.”
In their experiment, the scientists used a tip with a heat source about 20 nanometers high hovering between zero and 50 nanometers above a surface. The heat transfer occurs when cold molecules in the air are heated when they come in contact with the hot tip, then fly to the disk surface, colliding sometimes with other molecules before reaching the surface. Using Boltzmann’s law on the movement of gas, the scientists proposed for the first time a heat distribution at this scale and levels of heat flux.
The team showed that the heat transfer through the air takes only a few tens of picoseconds (10-12) seconds) to propagate from the tip to the surface when there is no contact. The scientists also found that below the height of 10 nanometers, a hot tip can heat a region with an edge of 35 nanometers while maintaining its shape; beyond this height, the shape is lost and the thermal spot increases significantly.
Heat-assisted data storage products, which are generally predicted to break into the market around 2010, could achieve data density of trillions of bits (terabytes) per square inch, dwarfing the current density. Scanning thermal microscopy, which acts like a nano thermometer to sense temperature and thermal conductivity on the nanoscale, could also benefit from this non-contact, localized heat transfer method.
“In thermally-assisted data storage, levels of heat flux are very important because you need to know if the increase of temperature is enough to reach the crtitical temperature (e.g. melting point),” said Sebastian Volz, coauthor of the study. “Scanning thermal microscopy can benefit from this work because it shows that, by reducing the heat source, you will probe more locally than what can be achieved now. Our work proposes a heat distribution in this field, which could help industrials design their devices.”
Citation: Chapuis, Pierre-Olivier, Greffet, Jean-Jacques, Joulain, Karl and Volz, Sebastian. Heat transfer between a nano-tip and a surface. Nanotechnology 17 (2006) 2978-2981.
By Lisa Zyga, Copyright 2006 PhysOrg.com.





















