New class of metal nitrides could lead to more durable semiconductors
New research shows that a novel class of nitrides made from noble metals can be synthesized under extreme conditions and are likely to have unusual or even unique properties that would be useful in semiconductor, superconductor and corrosion-resistant devices.
Historically, transition metal nitrides are fundamentally and technologically important because of their strength and durability, and are useful for their optical, electronic and magnetic properties. (“Nitride” is the name given to a nitrogen-containing compound in which nitrogen more strongly attracts the relevant electrons in the chemical bond.)
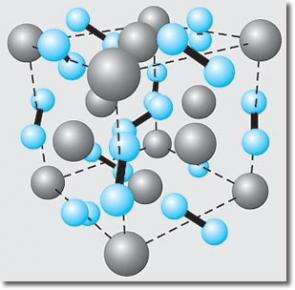
Using a diamond anvil cell to create high pressures and a laser to create high temperatures, scientists from Lawrence Livermore National Laboratory, in collaboration with researchers from the Carnegie Institution of Washington and the Atomic Weapons Establishment in England, created the first bulk nitride of the noble metal iridium. Noble metals are those that do not form compounds with other elements very easily. By combining experimental results with first-principle theoretical modeling, the scientists also have determined the structure of the known nitride of platinum as well as its bulk modulus (a measure of the material hardness). The results could prove useful to the semiconductor industry by making materials more durable and reliable.
“This work extends the scientific understanding of platinum and iridium nitrides,” said lead author Jonathan Crowhurst of Livermore’s Chemistry and Materials Science Directorate. “Demonstrating that these compounds exist and determining at least some of their physical properties should inspire the development of large-scale synthesis techniques to take advantage of their unusual properties,” Crowhurst said. “Platinum nitride, for example, has been shown to have a very high bulk modulus comparable to that of cubic boron nitride – a known super-hard material.”
The semiconductor industry currently uses titanium nitrides because of their strength and durability. The new nitrides may prove to be even more durable than titanium.
For platinum nitrides, synthesis conditions began at approximately 50 GPa (or 500,000 atmospheres of pressure) and 2,000 degrees Kelvin. Iridium nitrides were similar and would not occur below 47 GPa and 1,600 degrees Kelvin.
Further research is necessary to find a way to produce the nitrides industrially.
Other Livermore authors include Babak Sadigh, Cheryl Evans, James Ferreira and Art Nelson. The research, titled, “Synthesis and Characterization of the Nitrides of Platinum and Iridium,” appears in the March 3 edition of the journal Science.
Source: Lawrence Livermore National Laboratory