Genome sequencing aids investigation of an ancient and mysterious life-form
Researchers at the University of California, Santa Cruz, are using the latest in genetic technology to investigate an ancient form of life--the poorly understood microorganisms known as Archaea.
Many Archaea live in hostile environments, from salt lakes to acidic hot springs, but they can be very difficult to grow and study in the laboratory. So UCSC researchers were thrilled when their proposal to sequence the complete genomes of five species of Archaea was approved earlier this year by the Community Sequencing Program of the U.S. Department of Energy's Joint Genome Institute (JGI). JGI will carry out the sequencing and make the results publicly available.
"Having complete genome sequences will be enormously helpful to our research," said Todd Lowe, an assistant professor of biomolecular engineering.
Lowe and Chad Saltikov, an assistant professor of environmental toxicology at UCSC, teamed up with Sorel Fitz-Gibbon of UCLA and Christopher House of Pennsylvania State University to submit the Archaea proposal to the Community Sequencing Program. The five species they proposed for sequencing are closely related heat-loving Archaea found in various hot springs from Iceland to the Philippines. These species belong to a unique group of Archaea that can use sulfur compounds and toxic metals like arsenic and selenium to power their cells. They use these compounds in much the same way that our own cells use oxygen, Lowe said.
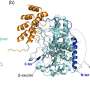
One of Lowe's goals is to identify the genes and metabolic pathways that enable the Archaea to thrive at extremely high temperatures and make use of toxic compounds. His lab investigates the unique biology of Archaea using a combination of laboratory experiments and computational techniques for genetic analysis.
Superficially, Archaea look much like bacteria. It wasn't until about 25 years ago that genetic techniques enabled scientists to see beyond the cell surface and recognize the differences between Archaea and bacteria.
"Archaea are as different from bacteria as bacteria are from us," Lowe said. "They account for a third of the biodiversity on the planet and they've hardly been studied."
Scientists now divide all known organisms into three distinct domains: the eukaryotes, which include all plants, animals, and fungi; the bacteria; and Archaea.
"We're studying a third group of life that we know very little about and that may tell us about the origin of life, how life works, and geochemical processes. It's really exciting," said Aaron Cozen, a graduate student in Lowe's lab.
Archaea have already yielded some useful products, such as heat-resistant enzymes used in laundry detergents. Other practical benefits are likely to emerge from ongoing research on the unusual metabolism and genetics of these organisms, said Saltikov, who is focusing on their metabolic use of arsenic.
The use of naturally occurring arsenic by Archaea and certain types of bacteria has serious consequences in some parts of the world, because the process converts the arsenic to a more toxic form that is also more likely to remain dissolved in water. In Bangladesh, many people have been poisoned by drinking water that is laden with this toxic form of arsenic. Saltikov and others have already found the bacterial gene involved in arsenic respiration, but they have not found a match for this gene in Archaea.
"Archaea may have the same gene, but the sequence is so different that it's not recognizable. Or they may have a completely different metabolic pathway, or an older pathway that leads us to greater understanding of the bacterial mechanism," Saltikov said.
For now, the biggest challenge for Lowe and Saltikov is learning how to grow Archaea and experiment with them in the lab. Many Archaea are poisoned by oxygen and have to be grown in special airtight containers. The heat-loving Archaea that Lowe is studying can only live at near-boiling temperatures and have to be grown in ovens. Keeping the conditions just right can be difficult and expensive, which limits the kinds of experiments researchers can do.
"These Archaea are more difficult than bacteria to grow and keep happy, so there are fewer ways we can assault them to learn about how they respond," Lowe said. He hopes that at least one of the five species chosen for genome sequencing will be easy to grow so it can become a model system for him and other scientists studying Archaea.
DNA microarrays (sometimes called "gene chips") are among the most powerful tools that Lowe is using to study Archaeal biology. Microarrays can provide a comprehensive view of gene activity in cells, revealing which genes are turned on and off in response to different conditions. Lowe's lab has already developed DNA microarrays based on the full genome sequences of two species of Archaea, and he plans to do the same for the five new species that will be sequenced by JGI. Using this "whole genome" approach to study Archaeal biology, he hopes to uncover the secrets that enable these organisms to live at the extreme limits of life on Earth.
"It's really exciting to know that you are the first person to understand something," Lowe said. "It's the potential for discovery of new biology that keeps me in this field."
Source: University of California, Santa Cruz