Biologists search for 'half-fusion'
Every living cell is surrounded by a membrane, a thin barrier that separates the genetic machinery of life from the non-living world outside. Though barriers, membranes are not impervious. Cells use a complex hierarchy of proteins that work in concert to allow cell membranes to fuse - with other cells or with membrane-encased packages of proteins and other chemicals that the cell needs to take in or release.
Though well-studied, the molecular details of membrane fusion remain mysterious. In particular, scientists don't understand how holes form between two membranes, but a new study by biochemists at Rice University and Iowa State University offers intriguing new clues about the nature of this process. The study is published in this month's issue of Nature Structural and Molecular Biology.
"Membrane fusion is one of the most basic processes of life," said James McNew, assistant professor of biochemistry and cell biology at Rice University. "It begins at fertilization and occurs billions of times a second in our bodies, and if it ever stops, we die."
For example, inside the cells in our brains, spines and nerves, membranes are used to seal up and transport tiny packets of signaling chemicals from the center of the cell to the outer cell membrane. These packets, or vesicles, wait just inside the cell membrane for the appropriate signal, and once they receive it, they fuse with the membrane and eject their contents into the surrounding tissue, causing an immediate chain reaction that keeps our hearts beating and allows us to move our muscles. Membrane fusion is also used to initiate disease.
"Some invading organisms like enveloped viruses use the fusion process to infect the cell," McNew said.
To understand membrane fusion, it helps to envision the basic structure of membranes. Just five billionths of meter across, membranes are bilayers, meaning they contain two separate layers, or sheets of fatty acids. Each of these sheets has a one side that is strongly attracted to water and one side that strongly repels it. The water-hating sides of the sheets stick tightly to one another, sealing out water on either side of the bilayer.
Additionally, all biological membranes are dotted with proteins, and some of these are called transmembrane proteins, meaning parts of them penetrate through the membrane like a needle through cloth. A large body of evidence suggests that a class of transmembrane proteins called SNAREs are responsible for driving membrane fusion during normal cellular activity. Exactly how they do this is unknown, but previous studies have suggested two possibilities.
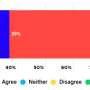
One model proposes that the portion of the SNARE protein that crosses the membrane forms a pore-like connection that mixes both layers of the membrane in one step. The other theory suggests that the SNARE proteins mix the two separate layers of a membrane one at a time, generating an intermediate stated called "hemifusion" or half-fusion. During hemifusion, the outer, water-loving sides of two membranes become connected, and the inner water-loving layers do not. In this state, the combining cells or vesicles could transfer proteins and other material stuck to their outside layers, but they do not exchange any material that's locked inside. Hemifusion has been observed in non-biological membranes containing no proteins, but has been difficult to detect with SNARE proteins.
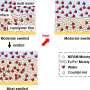
McNew and his Iowa State colleagues, Yeon-Kyun Shin, Zengliu Su, Fan Zhang and Yibin Xu, developed an ingenious method of tagging both inner and outer portions of the synthetic membranes with fluorescent dyes so they could use fluorescence spectroscopy to assay mixing of the inner and outer layers.
McNew and colleagues sought to find out if hemifusion was an intermediate fusion state in biological systems, so they created a test system that contained a lipid bilayer studed with SNARE proteins taken from bakers yeast. Using both normal SNAREs and a mutant variety, they were able to show that membrane fusion catalyzed by the SNARE machinery mixes the outer layer of the membrane separately from the inner layer -- a hallmark of hemifusion -- suggesting that a hemifusion intermediate can exist in biological systems and may well be the mechanism that all living cells utilize.
Preliminary data from follow-up studies indicate that these results are also generalizable to SNARE proteins from animals.
Source: Iowa State University